Tagged fda
ASTCT Supports the FDA’s Approval of Earlier Use of CAR T-Cell Therapy for Multiple Myeloma
The Food and Drug Administration approves CAR T-Cell therapy Carvykti® as a second-line treatment and the CAR T-Cell therapy Abecma® as a third-line treatment for adult patients with multiple myeloma.
Read Press Release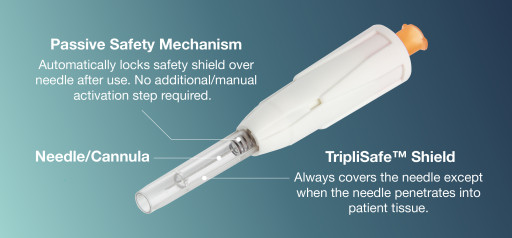
Amid FDA Concerns, HTL-Strefa Inc. Offers a Reliable and Safe Alternative to Chinese Supplied Needles & Syringes With European-Produced Sicura™ Passive Safety Needle
DropSafe® Sicura™ is the first and only fully passive safety needle in North America. Sicura™ designed to eliminate needlestick injuries, providing a safe injection experience, and is distributed by HTL-Strefa Inc., an MTD company.
Read Press Release
Amid FDA Concerns of Chinese Supplied Needles & Syringes, HTL-Strefa Inc. Offers a Reliable and Safe Alternative With Its European-Produced Passive Safety Needle, Sicura TM
DropSafe® Sicura™ is the first and only fully passive safety needle in North America. Sicura™ is designed to eliminate needlestick injuries, providing a safe injection experience, and is distributed by HTL-Strefa Inc., an MTD company.
Read Press Release
ZetrOZ Systems’ sam 2.0 Wearable Ultrasound Unit Receives New FDA Clearance for Expanded Medical Indications
Agency’s decision signifies that ZetrOZ’s sustained acoustic medicine technology is safe and effective for promoting injury healing in wider range of medical conditions.
Read Press Release
Selux Diagnostics Receives FDA Clearance on Their Gram-Negative Panel, Expanding the Antibiotic Menu for Its Next Generation Phenotyping System for Rapid AST
Approval gives way to the most comprehensive single menu offered by an AST platform while delivering precise, rapid results in 5.5 hours from isolated colonies.
Read Press Release
FDA Clears 'Selux NGP System' for Rapid Antibiotic Susceptibility Testing
In vitro antimicrobial resistance test enables clinical labs to deliver targeted therapeutic results days faster than the current standard of care, clearing the path for personalized antibiotic therapies
Read Press Release
Senzo Secures Additional $1.9MM Equity Funding for Its PCR-Accurate Lateral Flow Test
The funding, led by existing investors BioAdvance and Wellness Coaches, follows additional 3rd party studies by University College London and LateralDx, confirming that Senzo's Amplified Lateral Flow (ALF) platform returned greater than 98% accuracy against PCR, including in low viral-load samples
Read Press ReleaseRecardio and FDA Reach Agreement on Pivotal Phase 3 Trial Design
Recardio Inc., a late stage clinical-stage life science company developing regenerative therapies for cardiovascular diseases, announced that the FDA concurs with Recardio's pivotal Heal-MI Phase 3 trial design with Dutogliptin in Acute Myocardial Infarction.
Read Press Release
Caddo Mountain Spring Water, LLC Announces Plans for a New $24 Million Premium Water Bottling Facility in the Caddo Mountains of Arkansas
Company's Premium Water Bottling Facility will be State-of-the-Art. The Company's Plant will be a U.S. Advanced Manufacturing Plant by virtue of its design, engineering, and sustainability.
Read Press Release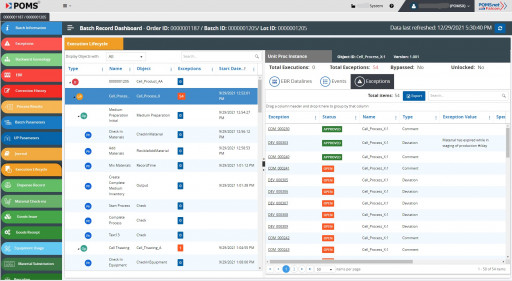
POMSnet Falcon Dives Into Pharma and Biotech GMP Facilities Across the Globe
POMS Corporation unveils a game changer in MES Dashboards & Batch Records for Pharma, Biotech, and Cell and Gene manufacturers. POMSnet Falcon swoops in and shatters the paradigm of electronic batch record visualization with a stunning original design. Go faster and see farther with POMSnet Falcon.
Read Press Release
Bidi Vapor Forges Ahead With Product-Specific Studies to Meet FDA's Rigorous Public Health Standard for Marketing ENDS
E-cig maker committed to demonstrating BIDI® Stick ENDS is appropriate for the protection of the public health, flavored products provide added benefit for adult cigarette smokers while outweighing potential risk to youth
Read Press Release
VoluMetrix's NIVA|HF for Heart Failure Patients Designated as Breakthrough Device by U.S. FDA
NIVA|HF is an investigational device designed to monitor the venous waveform, a novel physiologic signal, in heart failure patients. As a Breakthrough Device, NIVA|HF will receive pre-market developmental assistance and prioritized regulatory review from FDA. NIVA|HF is the first application of VoluMetrix's proprietary venous waveform technology.
Read Press Release